Peptide Nucleic Acid (PNA) Synthesis
Peptide Nucleic Acid (PNA) is a synthetic analogue of DNA with a neutral peptide-like backbone made of N-(2-aminoethyl)glycine instead of sugar-phosphate units. This design gives PNA exceptional chemical and enzymatic stability, allowing it to remain intact inside cells. PNAs bind complementary DNA or RNA sequences with high affinity through Watson–Crick base pairing, forming thermally stable hybrids and triplex structures by strand invasion. Their robust stability and precise hybridization make PNAs powerful tools for molecular diagnostics and antisense therapeutic research.
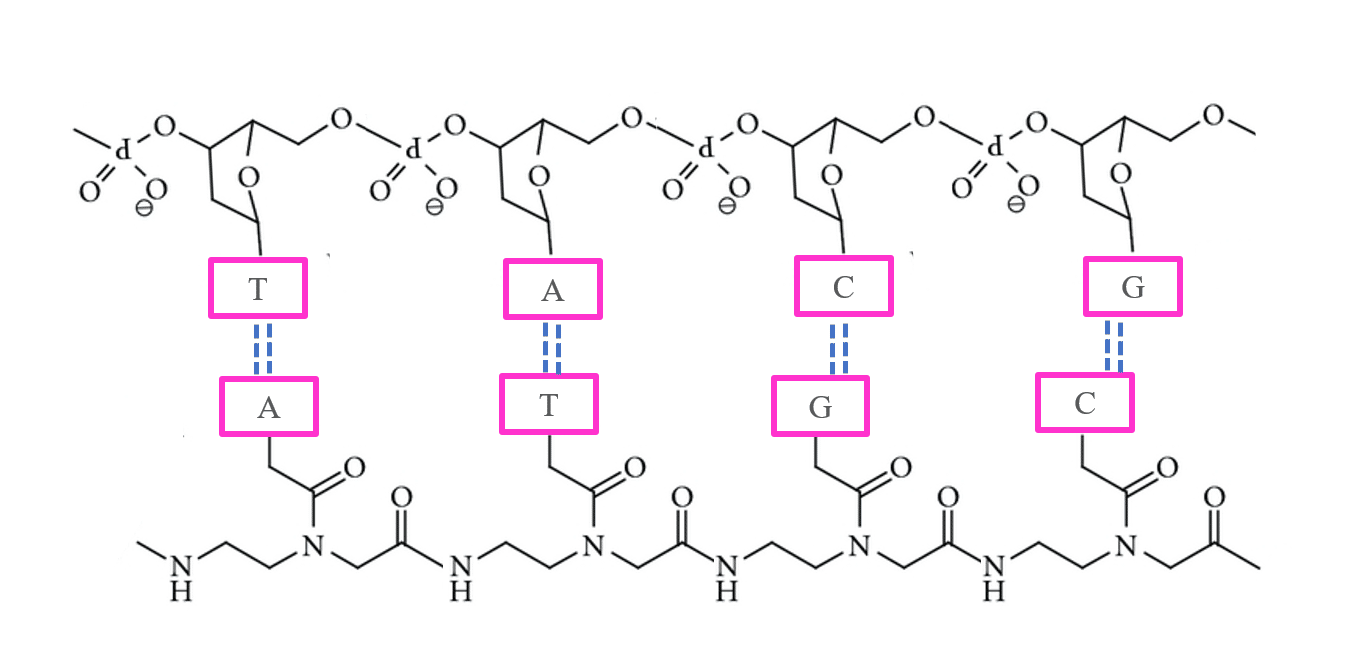
Peptide Nucleic Acid Structure
How is PNA different from DNA?
In peptide nucleic acids (PNAs), the usual phosphodiester backbone of DNA is substituted with repeating N-(2-aminoethyl)glycine units, to which purine and pyrimidine bases are attached through a methylene carbonyl linker.
At SB Peptide, we specialize in high-quality PNA synthesis using Fmoc-based solid-phase peptide synthesis (SPPS), ensuring excellent purity, consistency, and reproducibility. Our custom PNA services include the incorporation of a wide range of functional modifications, such as fluorophores, biotin, and other molecular tags, to support applications in molecular detection, diagnostics, and nucleic acid binding studies. Current developments in the field focus on advanced PNA designs, including hybrid PNA–peptide structures, which enhance solubility, improve cellular uptake, and expand biological functionality for research and therapeutic applications.
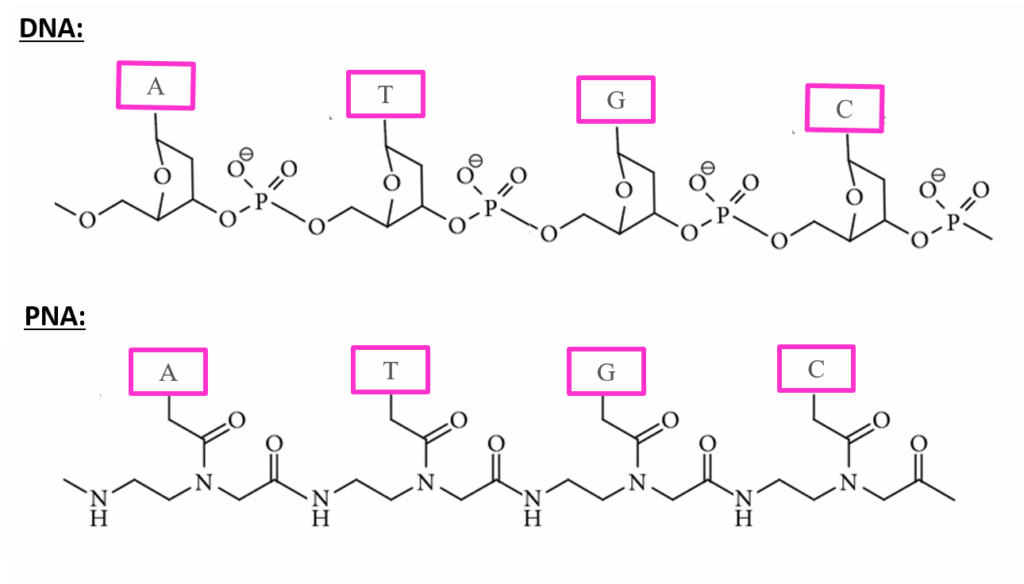
| FEATURES | PNA | DNA |
| Chemical stability | Stable over a wide pH range | Less stable in strongly acidic or basic media |
| Biological stability | Highly resistant to nucleases and most enzymes | Readily degraded by nucleases |
| Solubility | Moderate (can be improved by simple modifications) | Generally good aqueous solubility |
| Salt dependence for hybridization | Largely independent of salt concentration | Strongly dependent on ionic strength |
| ΔTm for single-base mismatch | Often <10 °C drop in Tm | Usually <15 °C drop in Tm |
| Binding affinity | Typically ≥1 °C higher Tm per base than DNA/DNA duplexes | - |
| Hybridization rate | Around 100–1000 times faster than DNA | - |
| Typical probe length | Commonly 13–18 bases | Commonly 25–30 bases |
PNA Technology
Advantages of PNA
- High Chemical Stability: Resistant to oxidation and hydrolysis, ensuring long-lasting performance.
- Enzymatic Protection: The neutral backbone prevents degradation by nucleases and proteases.
- Accurate Sequence Recognition: Binds specifically to complementary DNA or RNA targets.
- Stable Hybrid Formation: PNA/DNA and PNA/RNA complexes show strong thermal stability.
- Customizable Design: Easily tailored for gene detection, labeling, or antisense applications.
- Broad Research Use: Ideal for diagnostics, in situ hybridization, and gene modulation studies.
Properties of PNA
High Binding Affinity
PNAs form exceptionally stable duplexes with complementary DNA or RNA strands. Their neutral backbone eliminates electrostatic repulsion, while the planar base arrangement enhances stacking interactions, resulting in superior binding strength. Thanks to this remarkable affinity and precision, PNAs are highly effective for mutation detection, gene analysis, and other applications that demand strong, specific hybridization compared to natural oligonucleotides.
Resistance to Enzymatic Degradation
PNAs are not recognized by nucleases or proteases, making them highly resistant to enzymatic breakdown. This exceptional stability ensures reliable performance in biological environments, allowing prolonged interaction with target nucleic acids and improved accuracy in diagnostic assays.
Sequence Specificity
PNAs display exceptional sequence discrimination, capable of distinguishing even a single-base difference in target DNA or RNA. This high precision enables reliable detection of mutations and single nucleotide polymorphisms (SNPs), as a single mismatch significantly reduces the stability of the PNA–DNA duplex, enhancing accuracy in genetic analysis and diagnostics.
Neutral Charge
Unlike DNA or RNA, PNAs possess a neutral backbone that eliminates the need for salt to stabilize hybrid formation. This allows PNAs to hybridize efficiently even under low-salt conditions, which can be beneficial in various biological and experimental contexts. Their lack of negative charge also minimizes nonspecific binding to other biomolecules, resulting in more accurate and selective nucleic acid targeting.
PNA Applications
What are PNAs used for?
FISH Probes
Molecular Diagnostics
Antisense Research
Gene Detection
Therapeutic Development
Biomarker Discovery
Genetic Analysis
Pathogen Detection
Cancer Research
Epigenetic Studies
RNA Targeting
In Situ Hybridization
Gene Expression Profiling
Mutation Analysis
Drug Development
Synthetic Biology
Nanotechnology
Forensic Science
What we offer at SB Peptide
Flexible Synthesis Scales
Standard sizes for custom PNA synthesis are 20 nmol, 50 nmol, 100 nmol and 200 nmol. Bulk quantities are available upon requests. Standard purity rate is 90%.
Available PNA Modifications
A wide range of PNA modifications are available at the 5′ or 3′ ends, including:
PNA-CPP
Conjugation to CPPs (cell penetrating peptides)
PNA-DNA/RNA
Conjugation to DNA/RNA
Fluorescent-PNA
Conjugation to a wide range of fluorescent dyes (fluorescein, rhodamine, cyanines)
Biotinylated-PNA
Conjugation to biotin
Alkyne/azide-PNA
Incorporation of alkyne or azide function (click chemistry)
Other modification
Linkers and Solubility Optimization
Linkers are commonly used to connect PNA sequences with functional groups such as peptides, fluorophores, or other molecules, while improving overall performance. Hydrophilic and flexible linkers can significantly enhance solubility, reduce aggregation, and improve probe behavior, especially for longer or purine-rich PNAs.
At SB Peptide, we offer a selected set of linkers that can be introduced at the C-terminus (3′-end) to optimize PNA performance and conjugation:
AEEA (eg1)
PEG-type spacer that improves flexibility, increases solubility, and enhances fluorescence performance by reducing quenching
Glycine (Gly)
A short and flexible spacer providing minimal distance with low structural impact
Lysine (Lys)
It introduces positive charge to improve solubility, reduce aggregation, and enhance interaction with nucleic acids
Design Considerations
To ensure optimal PNA solubility and performance, we recommend following these design guidelines:
- Maintain length below ~30 bases
- Keep purine content under 50%
- Keep guanine content under 35%
- Limit purine stretches (>6–7 bases)
- Minimize self-complementarity (>4–5 bases)
Following these guidelines ensures excellent solubility, particularly in aqueous environments.
For applications such as PCR clamping, it is recommended to optimize the sequence or include solubility-enhancing modifications (e.g. Lys or AEEA linkers). In applications involving organic solvents (e.g. FISH), these limitations are generally less critical.
Additional strategies to improve solubility include:
- Use of formamide or DMSO in solution
- Mild heating during dissolution
Custom PNA Synthesis Services
PNA Library Synthesis
SB-PEPTIDE can produce libraries of hundreds of customized PNA in parallel. Modifications are also available with this service.
Express PNA Synthesis
Standard production takes 3–4 weeks, but for tight deadlines, a 12-day express synthesis service is available for custom PNA.
Quote Request
Contact us for bulk pricing or custom specifications. Our team will help you determine the optimal scale for your project.
European Research Collaboration
SB Peptide participates in international research initiatives supporting the development of innovative biomolecules for biomedical applications.
We collaborate with the NAV15-CARED European consortium contributing expertise in peptide synthesis and molecular engineering for advanced therapeutic research.
References
- Brodyagin, N. et al. Peptide nucleic acids: structure, properties, and applications. Molecules, 2021. https://pmc.ncbi.nlm.nih.gov/articles/PMC8313981/
- Quijano, E. et al. Peptide nucleic acids: an overview of chemical and biological properties. Molecules, 2018. https://pmc.ncbi.nlm.nih.gov/articles/PMC5733847/
- Pipkorn, R. et al. Advances in Fmoc-based solid-phase synthesis of PNA and conjugates. Molecules, 2011. https://pmc.ncbi.nlm.nih.gov/articles/PMC3222083/
- Pellestor, F. et al. Peptide nucleic acids (PNA): a tool for diagnostics and cytogenetics. European Journal of Medical Genetics, 2004. https://www.nature.com/articles/5201226
- Jiang, X. et al. PNA-based probes for nucleic acid detection and diagnostics. Analytical Chemistry, 2023. https://pubs.acs.org/doi/10.1021/acs.analchem.3c01809
- Pellestor, F. et al. Applications of PNA probes in FISH for chromosome and telomere analysis. European Journal of Medical Genetics, 2004. https://www.nature.com/articles/5201226
- Meindl, C. et al. PNA-based detection of bacterial pathogens in complex samples. Biosensors and Bioelectronics, 2026. https://www.sciencedirect.com/science/article/pii/S0956566326001132
- Tareum, S. et al. Validation of a PNA clamping method to reduce host DNA amplification in microbiome studies. Phytobiomes Journal, 2020.
- Hu, J. et al. Antisense peptide nucleic acids for gene regulation. Proceedings of the National Academy of Sciences (PNAS), 2007. https://pmc.ncbi.nlm.nih.gov/articles/PMC2564818/
- Brodyagin, N. et al. Structural design of PNAs and role of lysine in solubility enhancement. Molecules, 2021. https://pmc.ncbi.nlm.nih.gov/articles/PMC8313981/
- Tsourkas, A. et al. Impact of lysine conjugation on PNA binding and stability. ACS Omega, 2020. https://pubs.acs.org/doi/10.1021/acsomega.0c04021
- Tsylentis, P. et al. Peptide nucleic acid conjugates and antimicrobial applications. https://pmc.ncbi.nlm.nih.gov/articles/PMC10618302/
PNA Synthesis - Custom support & Price inquiry
Address
Reach out to us for a personalized quote or to learn more about our PNA products and services. Visit us at 3b Rue de l'Isère
38120 Saint Egrève, FRANCE
Phone
+33 (0) 4 76 45 21 20
