MAGE-A3 (112-120) peptide – KVAELVHFL – Epitope of Melanoma Antigen Gene A3
SB-PEPTIDE offers MAGE-A3 (112-120) HLA-A*02:01 KVAELVHFL T-cell epitope for antigen-specific T-cells stimulation in ELISPOT,ICS,cytotoxity or proliferation assays.
MAGE-A3 protein
MAGE-A3 (UniProt: P43357) is a tumor-specific protein expressed in more than 70% of metastatic melanomas,as well as a wide range of non-melanoma epithelial malignancies including lung,ovarian,bladder,head and neck carcinomas,multiple myeloma and hepatocellular carcinoma.
MAGE-A3 shares 73% identity with MAGE-A1 protein (UniProt: P43355) and is recognized by autologous cytolytic T-lymphocytes (CTL).
KVAELVHFL – HLA-A*02:01 T-cell epitope
To stimulate antigen-specific T-cells,peptides are presented by major histocompatibility complex (MHC) class I molecules on the surface of antigen-presenting cells (APCs). Usually,antigenic peptides are between 8 and 11 amino acids in length and exhibit MHC-specific sequence motifs.
HLA-A*02:01 restricted KVAELVHFL epitope derives from MAGE-A3 protein proteolysis in the cytosolic compartment. After digestion,Peptide-MHC-I complexes are transported to the surface of APCs where they will be recognized by CD8+ CTL. This interaction will activate CTL and initiate their differentiation to ensure abnormal cells elimination.
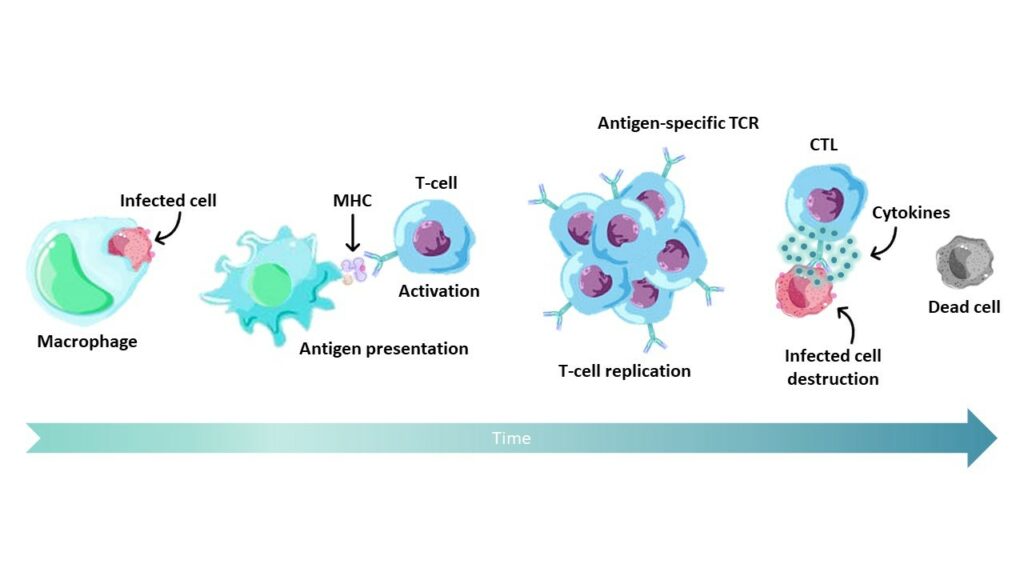
Among MAGE cancer testis antigens (CTA),MAGE-A3 antigenic peptides represent attractive candidates towards targeted adoptive immunotherapy of cancer.
Order SB-PEPTIDE’s KVAELVHFL epitope today to develop the next T-cell immunotherapy that will help save many patients’ lives !
Technical specification
 |
Sequence : KVAELVHFL |
 |
MW : 1055 ,27 g/mol (C51H82N12O12) |
 |
Purity : > 95% |
 |
Counter-Ion : TFA Salts (see option TFA removal) |
 |
Delivery format : Freeze dried in propylene 2mL microtubes |
 |
Peptide Solubility Guideline |
 |
Bulk peptide quantities available |
Price
| Product catalog | Size | Price € HT | Price $ HT |
| SB306-1MG | 1 mg | 88 | 110 |
| SB306-5MG | 5 mg | 308 | 385 |
| SB306-10MG | 10 mg | 522 | 653 |
| SB306-50MG | 50 mg | 1815 | 2268 |
References
J Immunol. 2011 Jan 15;186 (2):685–696. doi: https://doi.org/10.4049/jimmunol.1001775
Human gene MAGE-3 codes for an antigen recognized on a melanoma by autologous cytolytic T lymphocytes.
Human melanoma cell line MZ2-MEL expresses several antigens recognized by autologous cytolytic T lymphocyte (CTL) clones. We reported previously the identification of a gene,named MAGE-1,that codes for one of these antigens named MZ2-E. We show here that antigen MZ2-D,which is present on the same tumor,is encoded by another member of the MAGE gene family named MAGE-3. Like MAGE-1,MAGE-3 is composed of three exons and the large open reading frame is entirely located in the third exon. Its sequence shows 73% identity with MAGE-1. Like MZ2-E,antigen MZ2-D is presented by HLA-A1. The antigenic peptide of MZ2-D is a nonapeptide that is encoded by the sequence of MAGE-3 that is homologous to the MAGE-1 sequence coding for the MZ2-E peptide. Competition experiments using single Ala-substituted peptides indicated that amino acid residues Asp in position 3 and Tyr in position 9 were essential for binding of the MAGE-1 peptide to HLA-A1. Gene MAGE-3 is expressed in many tumors of several types,such as melanoma,head and neck squamous cell carcinoma,lung carcinoma and breast carcinoma,but not in normal tissues except for testes. It is expressed in a larger proportion of melanoma samples than MAGE-1. MAGE-3 encoded antigens may therefore have a wide applicability for specific immunotherapy of melanoma patients.
Cell - Cancer Immunotherapy. 2010 May;18(1):S72-S73. doi: https://doi.org/10.1016/S1525-0016(16)37631-6
190. Development of an HLA-A*0201 Restricted T Cell Receptor That Recognizes Peptide Epitopes from MAGE-A3 and MAGE-A12 for Targeted Adoptive T Cell Immunotherapy of Multiple Cancer Types
Adoptive cell transfer therapy using peripheral blood lymphocytes (PBL) genetically engineered to express tumor antigen-specific T cell receptors (TCRs) is a promising strategy for treating patients with malignancies. TCR engineered PBL targeting melanocyte differentiation antigens mediated objective tumor regression in patients with metastatic melanoma. However,this therapy has also been shown to lead to severe on-target toxicity against skin,eye and ear,normal tissue that contain antigen-expressing melanocytes (Johnson LA et al,Blood,114: 535–546,2009). In an attempt to avoid toxicity to normal tissue,TCRs were derived that recognize cancer testis antigens (CTA). These antigens are expressed in multiple tumors with no expression in normal tissue with the exception of immunoprivileged sites such as testis and placenta that lack expression of MHC class I molecules. The MAGE family of CTA represent attractive candidates for targeted adoptive immunotherapy of cancer and one of the members of this family MAGE-A3 is expressed in >70% of metastatic melanomas,as well as a wide range of non-melanoma epithelial malignancies including lung,ovarian,bladder,head and neck carcinomas,multiple myeloma and hepatocellular carcinoma…
J Exp Med. 1994 Mar 01;179(3):921–930. doi: https://doi.org/10.1084/jem.179.3.921
A TCR Targeting the HLA-A*0201–Restricted Epitope of MAGE-A3 Recognizes Multiple Epitopes of the MAGE-A Antigen Superfamily in Several Types of Cancer
Adoptive immunotherapy using TCR-engineered PBLs against melanocyte differentiation Ags mediates objective tumor regression but is associated with on-target toxicity. To avoid toxicity to normal tissues,we targeted cancer testis Ag (CTA) MAGE-A3,which is widely expressed in a range of epithelial malignancies but is not expressed in most normal tissues. To generate high-avidity TCRs against MAGE-A3,we employed a transgenic mouse model that expresses the human HLA-A*0201 molecule. Mice were immunized with two HLA-A*0201–restricted peptides of MAGE-A3: 112–120 (KVAELVHFL) or MAGE-A3: 271–279 (FLWGPRALV),and T cell clones were generated. MAGE-A3–specific TCR α- and β-chains were isolated and cloned into a retroviral vector. Expression of both TCRs in human PBLs demonstrated Ag-specific reactivity against a range of melanoma and nonmelanoma tumor cells. The TCR against MAGE-A3: 112–120 was selected for further development based on superior reactivity against tumor target cells. Interestingly,peptide epitopes from MAGE-A3 and MAGE-A12 (and to a lesser extent,peptides from MAGE-A2 and MAGE-A6) were recognized by PBLs engineered to express this TCR. To further improve TCR function,single amino acid variants of the CDR3 α-chain were generated. Substitution of alanine to threonine at position 118 of the α-chain in the CDR3 region of the TCR improved its functional avidity in CD4 and CD8 cells. On the basis of these results,a clinical trial is planned in which patients bearing a variety of tumor histologies will receive autologous PBLs that have been transduced with this optimized anti–MAGE-A3 TCR.
