SAHM1
Cancer Signalling Peptides
SAHM1 is a stapled peptide mimetic derived from a dominant-negative form of Mastermind-like (MAML) that disrupts formation of Notch transcriptional complex. It is widely used as a research tool to investigate Notch signaling and allergic airway inflammation in murine models.
Diseases Target
T-cell acute lymphoblastic leukemia (T-ALL), triple-negative breast cancer (TNBC), and allergic airway inflammation including asthma.
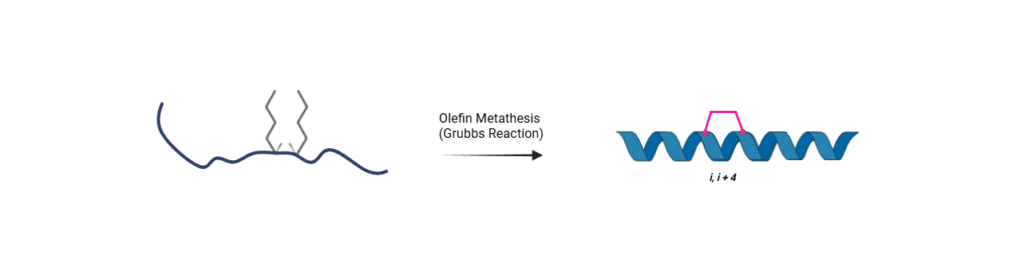
Stapling Strategy
Fmoc-(S)-2-(4-pentenyl)alanine (Fmoc-(S5)-OH, CAS: 288617-73-2) enables the solid-phase peptide synthesis (SPPS) of SAHM1. During assembly, Fmoc-(S5)-OH is incorporated at the i,i+4 positions (9,13), followed by ruthenium-catalyzed ring-closing olefin metathesis to generate the (CH₂)₄ hydrocarbon staple.
Technical specification
 |
Sequency : | Ac-βAla-ERLRRRI(S5)LCR(S5)HHST-NH₂ |
 |
MW : | 2195.60 g/mol |
| Purity : | > 95% | |
| Counter-Ion : | TFA Salts | |
| Delivery format : | Lyophilized | |
| CAS number: | 2050906-89-1 |
Price
| Product | Size | Price € |
Price $ |
| SB324-1mg | 1 mg | 601 | 722 |
| SB324-5mg | 5 mg | 763 | 916 |
| SB324-10mg | 10 mg | 1029 | 1235 |
For more information about stapled peptides, please visit our dedicated page.
Explore our Stapled Peptide Catalog products. Click HERE to discover additional reference compounds.
Custom Stapled Peptide Services
If your target is not listed, we offer custom stapled peptide design and synthesis. Whether you require sequence optimization, specific modifications, or larger production quantities, our team can support your project. Submit your project details and we will provide a personalized proposal.
References
2009 Nov 12;462(7270):182-8. doi: 10.1038/nature08543.
Direct inhibition of the NOTCH transcription factor complex
Abstract
Direct inhibition of transcription factor complexes remains a central challenge in the discipline of ligand discovery. In general, these proteins lack surface involutions suitable for high-affinity binding by small molecules. Here we report the design of synthetic, cell-permeable, stabilized alpha-helical peptides that target a critical protein-protein interface in the NOTCH transactivation complex. We demonstrate that direct, high-affinity binding of the hydrocarbon-stapled peptide SAHM1 prevents assembly of the active transcriptional complex. Inappropriate NOTCH activation is directly implicated in the pathogenesis of several disease states, including T-cell acute lymphoblastic leukaemia (T-ALL). The treatment of leukaemic cells with SAHM1 results in genome-wide suppression of NOTCH-activated genes. Direct antagonism of the NOTCH transcriptional program causes potent, NOTCH-specific anti-proliferative effects in cultured cells and in a mouse model of NOTCH1-driven T-ALL.
2015 Nov;116(11):2598-609. doi: 10.1002/jcb.25205.
Notch signaling promotes osteoclast maturation and resorptive activity
Abstract
The role of Notch signaling in osteoclast differentiation is controversial with conflicting experimental evidence indicating both stimulatory and inhibitory roles. Differences in experimental protocols and in vivo versus in vitro models may explain the discrepancies between studies. In this study, we investigated cell autonomous roles of Notch signaling in osteoclast differentiation and function by altering Notch signaling during osteoclast differentiation using stimulation with immobilized ligands Jagged1 or Delta-like1 or by suppression with γ-secretase inhibitor DAPT or transcriptional inhibitor SAHM1. Stimulation of Notch signaling in committed osteoclast precursors resulted in larger osteoclasts with a greater number of nuclei and resorptive activity whereas suppression resulted in smaller osteoclasts with fewer nuclei and suppressed resorptive activity. Conversely, stimulation of Notch signaling in osteoclast precursors prior to induction of osteoclastogenesis resulted in fewer osteoclasts. Our data support a mechanism of context-specific Notch signaling effects wherein Notch stimulation inhibits commitment to osteoclast differentiation, but enhances the maturation and function of committed precursors.
2018 Jul;142(1):76-85.e8. doi: 10.1016/j.jaci.2017.08.042. Epub 2017 Oct 27.
The Notch pathway inhibitor stapled α-helical peptide derived from mastermind-like 1 (SAHM1) abrogates the hallmarks of allergic asthma
Abstract
Background: The Notch signaling pathway has been implicated in the pathogenesis of allergic airway inflammation. Targeting the active Notch transactivation complex by using the cell-permeable, hydrocarbon-stapled synthetic peptide stapled α-helical peptide derived from mastermind-like 1 (SAHM1) resulted in genome-wide suppression of Notch-activated genes in leukemic cells and other models. However, the efficacy of SAHM1 in allergic asthma models has remained unexplored.
Objective: We aimed to investigate the therapeutic efficacy of SAHM1 in a house dust mite (HDM)-driven asthma model.
Methods:Topical therapeutic intervention with SAHM1 or a control peptide was performed during sensitization, challenge, or both with HDM in mice. Airway inflammation was assessed by using multicolor flow cytometry, and bronchial hyperreactivity was studied. Additionally, SAHM1 therapy was investigated in mice with established allergic airway inflammation and in a model in which we neutralized IFN-γ during HDM challenge to support the TH2 response and exacerbate asthma.
Results: SAHM1 treatment during the challenge phase led to a marked reduction of eosinophil and T cell numbers in bronchoalveolar lavage fluid compared with those in diluent-treated or control peptide-treated mice. Likewise, T-cell cytokine content and bronchial hyperreactivity were reduced. SAHM1 treatment dampened TH2 inflammation during ongoing HDM challenge and enhanced recovery after established asthma. Additionally, in the presence of anti-IFN-γ antibodies, SAHM1 downregulated expression of the key TH2 transcription factor GATA3 and intracellular IL-4 in bronchoalveolar lavage fluid T cells, but expression of the TH17 transcription factor retinoic acid-related orphan receptor γt or intracellular IL-17 was not affected. SAHM1 therapy also reduced serum IgE levels.
Conclusions: Therapeutic intervention of Notch signaling by SAHM1 inhibits allergic airway inflammation in mice and is therefore an interesting new topical treatment opportunity in asthmatic patients.
