BIM BH3 _ SAHB (Stabilized Alpha-Helix of BCL-2 domains)
Cancer Research Peptide
The BIM BH3 staple peptide is a hydrocarbon-stapled α-helical peptide derived from the BH3 domain of the pro-apoptotic BCL-2 family protein BID. This stapled peptide is designed to mimic the native BH3 α-helix and selectively interact with anti-apoptotic BCL-2 family members. The hydrocarbon stapling strategy enhances α-helical stability, protease resistance, and cellular uptake compared to the corresponding linear peptide. BIM SAHB is widely used as a research tool to study apoptosis regulation and protein–protein interactions involved in cancer biology.
Disease Target
Leukemias, lymphomas, multiple myeloma, and selected solid tumors such as lung carcinoma.
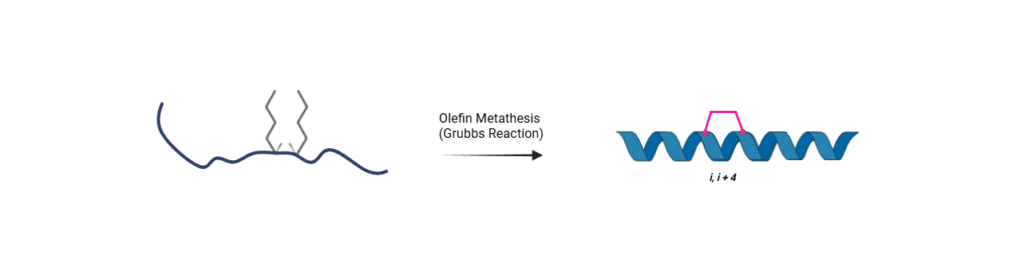
Stapling Strategy
SAHB (BID BH3) is synthesized using Fmoc-(S)-2-(4-pentenyl)alanine (Fmoc-(S5)-OH, CAS: 288617-73-2), incorporated at i,i+4 positions during solid-phase peptide synthesis (SPPS). A ruthenium-catalyzed ring-closing olefin metathesis generates the hydrocarbon staple, stabilizing the α-helical BH3 structure and enhancing proteolytic stability and cellular activity.
Technical specification
 |
Sequency : | EDIIRNIARHLA(S5)VGD(S5)NleDRSIW |
 |
MW : | 2713.1 g/mol |
 |
Purity : | > 95% |
 |
Counter-Ion : | TFA Salts |
 |
Delivery format : | Lyophilized |
Price
| Product | Size | Price € |
Price $ |
| SB321-1mg | 1 mg | 651 | 781 |
| SB321-5mg | 5mg | 825 | 990 |
| SB321-10mg | 10mg | 1109 | 1331 |
For more information about stapled peptides, please visit our dedicated page.
Explore our Stapled Peptide Catalog products. Click HERE to discover additional reference compounds.
Custom Stapled Peptide Services
If your target is not listed, we offer custom stapled peptide design and synthesis. Whether you require sequence optimization, specific modifications, or larger production quantities, our team can support your project. Submit your project details and we will provide a personalized proposal.
References
2004 Sep 3;305(5689):1466-70. doi: 10.1126/science.1099191.
Activation of Apoptosis in Vivo by a Hydrocarbon-Stapled BH3 Helix
Abstract
BCL-2 family proteins constitute a critical control point for the regulation of apoptosis. Protein interaction between BCL-2 members is a prominent mechanism of control and is mediated through the amphipathic α-helical BH3 segment, an essential death domain. We used a chemical strategy, termed hydrocarbon stapling, to generate BH3 peptides with improved pharmacologic properties. The stapled peptides, called “stabilized alpha-helix of BCL-2 domains” (SAHBs), proved to be helical, protease-resistant, and cell-permeable molecules that bound with increased affinity to multidomain BCL-2 member pockets. A SAHB of the BH3 domain from the BID protein specifically activated the apoptotic pathway to kill leukemia cells. In addition, SAHB effectively inhibited the growth of human leukemia xenografts in vivo. Hydrocarbon stapling of native peptides may provide a useful strategy for experimental and therapeutic modulation of protein-protein interactions in many signaling pathways.
Blood. 2006;108(11):711.https://doi.org/10.1182/blood.V108.11.711.711
Anti-Leukemic Potency of Stapled BH3 Helices Correlates with Their Capacity for Bifunctional Activation of Apoptotic Pathways
Abstract
Selective targeting of apoptosis in vivo is a promising pharmacologic strategy for subverting cancer. BCL-2 family protein interactions constitute a critical control point for the regulation of apoptosis. Whereas multidomain anti-apoptotic proteins such as BCL-2 guard against cell death, multidomain pro-apoptotic proteins such as BAX constitute a gateway to cell death through mitochondrial damage. The BH3-only proteins function as death sentinels situated throughout the cell, poised to transmit signals of cell injury to multidomain members. BH3-only proteins deliver death signals via their alpha-helical BH3 domains, which are either neutralized by anti-apoptotic proteins or delivered, directly or indirectly, to the mitochondrial executioners BAX and BAK. By inserting hydrocarbon “staples” into native BH3 peptide sequences, we have generated a chemical toolbox of stabilized alpha-helices of BCL-2 domains (SAHBs) to dissect apoptotic pathways and develop prototype therapeutics. We previously demonstrated that a stapled peptide corresponding to the BID BH3 domain is a helical, protease-resistant, and cell-permeable compound that binds to anti-apoptotic targets with high affinity and exhibits anti-leukemic activity in vitro and in vivo. Using our expanded panel of compounds, we find that a stapled BAD BH3 likewise displays high affinity binding to select anti-apoptotic targets; however, BID SAHB is uniformly more potent than BAD SAHB in inducing apoptosis of a panel of leukemia cell lines. To explore the molecular mechanism underlying the differential potencies of BID and BAD SAHBs, we evaluated a model in which select BH3-only proteins directly engage pro-apoptotic multidomain proteins to trigger mitochondrial apoptosis. We detect and measure, for the first time, direct binding between select SAHBs, such as BID, and BAX. The observed interaction between BID SAHB and BAX triggered functional activation of BAX in vitro, resulting in mitochondrial cytochrome c release and FITC-dextran release from liposomes. The specificity of the BID SAHB-BAX interaction and its biochemical consequences is highlighted by abrogation of BID SAHB activity by point mutation and by competitive binding to anti-apoptotic BCL-XL. We confirmed the physiologic relevance of our observed in vitro interaction between BID SAHB and BAX by their co-immunoprecipitation from BID SAHB-treated leukemia cells. In contrast, BAD SAHB does not bind or activate multidomain pro-apoptotic BAX. These data provide an initial mechanistic explanation for the relative potency of BID SAHB in activating leukemia cell apoptosis. Thus, bifunctional SAHBs that directly engage both pro- and anti-apoptotic multidomain proteins may be more robust pro-apoptotic therapeutics, compared to compounds that selectively target anti-apoptotic proteins.
