Peptides labelled with affinity tags
SB-PEPTIDE can conjugate and functionnalize peptides with many different compounds including affinity tags. Affinity-tags can be biotin, maleimide, alkyne and azide function or thiol functions. These functions serve for bioconjugations, in this case between a peptide and something else (another peptide, protein, a surface, a fluorescent dye, a DNA...).
Affinity tagged peptides applications
- Purification by affinity using appropriate columns
- Labeling for detection assay using a fluorescent streptavidine tag or an antibody anti-biotin
- Immobilization on a solid support
Affinity tags available
His-tag peptides :
SB-PEPTIDE can synthesize peptides with several histidines at C-ter or N-ter to serve as His-tag.
Biotinylated-peptides:
Biotin, also known as vitamin B8 or B7 is water soluble. Biotin is widely used in bioconjugation thanks to its high affinity for streptavidine a 52,8 kDa protein with a Kd in the range of 10(−14) mol/L. Biotinylated-peptide is widely used for ELISA or pull-down assays. As biotin-streptavidin forms a large complex (>50kDa) compared to a few kilodaltons peptide, we recommend especially for fluorescent labeling to opt-in for another strategy. SB-PEPTIDE can conjugate specifically one biotin per custom synthetic peptides in N-ter, C-ter or within the sequence.
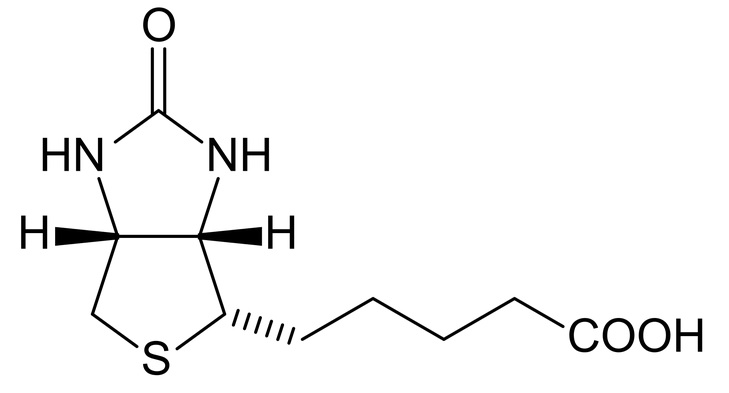
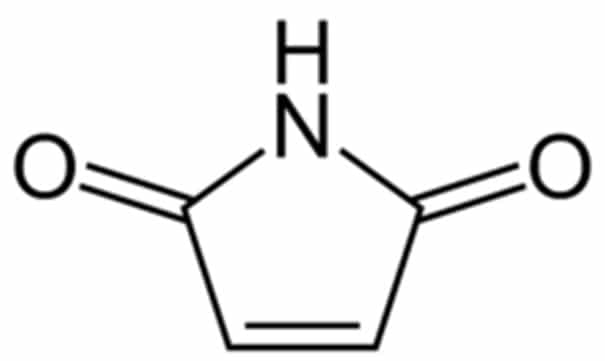
Maleimide-peptide:
SB-PEPTIDE can incorporate a maleimide function at N-ter, C-ter or inside the sequence. Maleimide can react with thio functions of cysteine to form a stable carbon-sulfur bond making it very strong and specific. Thiol-maleimide conjugation is widely used in bioconjugation thanks to its simplicity and attractive conjugation yield.
Azide/Alkyne-peptide:
Several azido and alkyne functions are available to prepare azido-peptide or alkyne-peptide suitable for click-chemistry. Click-chemistry is a reaction used in bioconjugation to specifically conjugate one compound azide-functionalized with another compound alkyne-functionalized. The classic click reaction is the copper-catalyzed reaction of an azide with an alkyne to form a 5-membered heteroatom ring: a Cu(I)-catalyzed azide-alkyne cycloaddition (CuAAC). Alkyne-peptides or azide-peptides are valuable approaches to consider for in-solution labeling when amide bonds or thiol-maleimide conjugations are not possible due to amino acids contained in the sequence.
Thiol-peptide:
Thiol-peptides are peptides containing a thiol function. Thiol function can be incorporated by adding a cysteine during the synthesis or other thiol existing thiolated compounds. Thiol-peptides are widely used for maleimide conjugation such as peptide-BSA or peptide KLH conjugation or for peptide attachment to gold surface (i.e. SPR).
Others upon request
Tags can be placed at various positions of the peptide such as N-ter, C-ter or within the peptide. N-terminal conjugation is generally the easier way, side chain labeling can also be performed. These functions are also availble for PNA custom synthesis.
Custom peptide synthesis
SB-PEPTIDE can custom synthesize peptides and functionalize them with affinity tags. Please get in touch for project evaluation.
