CENPB FAM-Ahx Probe
CENPB Pan-Centromere PNA FISH Probe
The CENPB FAM-Ahx probe is designed to recognize the CENP-B box, a conserved motif located within alpha-satellite DNA at human centromeres. This region plays a key role in centromere function, as it is involved in the binding of the CENP-B protein, which contributes to centromere structure and stability. The probe binds specifically to centromeric regions across human and mouse chromosomes, with the exception of the Y chromosome. The reverse complementary sequence (5' to 3') is tcccgtttccaacgaat.
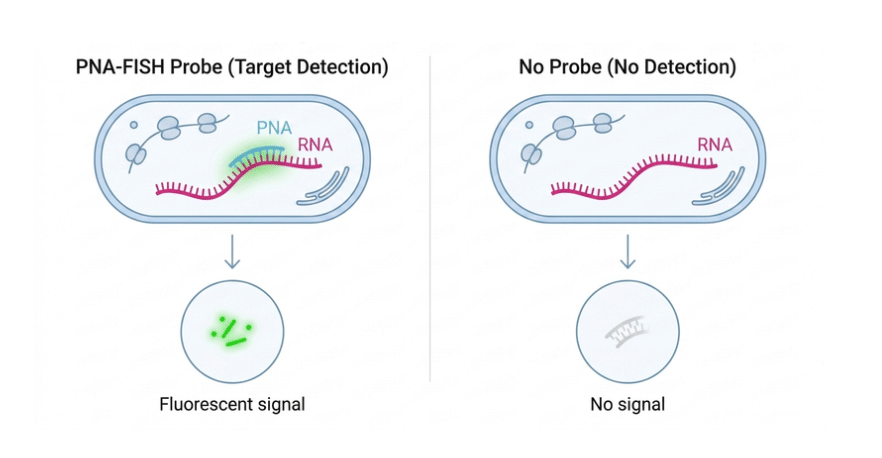
Main characteristics
The CENPB probe is commonly used in Fluorescence In Situ Hybridization (FISH) to detect and visualize centromeric regions by specifically recognizing the CENP-B box. This allows researchers to monitor centromere behavior during cell division and study chromosome segregation and structural integrity. Since CENP-B is essential for the organization of centromeric chromatin and proper kinetochore assembly, the probe is a valuable tool for analyzing chromosome dynamics during mitosis. The probe is conjugated with FAM (Ex/Em = 495/520 nm), allowing direct detection under fluorescence microscopy. An Ahx spacer (6-aminohexanoic acid, CAS: 60-32-2) is introduced to separate the fluorophore from the recognition sequence, improving accessibility and hybridization efficiency. Additionally, a terminal glycine may be incorporated into the PNA structure to increase molecular flexibility and support optimal binding performance without compromising specificity.
Applications
- Genome-wide identification of centromeric regions
- Detection of chromosomal abnormalities in oncology
- Screening for aneuploidies before and after birth
- Assessment of genomic stability in stem cell studies
- Analysis of chromosomal variations using hybridization techniques
- Detailed chromosome structure analysis at high resolution
Technical specification
 |
Sequency : | FAM-Ahx-attcgttggaaacggga-G |
 |
MW : | 5237.95 g/mol |
 |
Purity : | > 95% |
 |
Counter-Ion : | TFA Salts |
 |
Delivery format : | Lyophilized |
Price
| Product | Size | Price € |
Price $ |
| SB343 - 25nmol | 25 nmol | 499 | 599 |
| SB343 - 50 nmol | 50 nmol | 744 | 892 |
For more information about PNA, please visit our dedicated page.
Explore our PNA Catalog products. Click HERE to discover additional reference compounds.
Custom PNA Services
If your target is not listed, we offer custom PNA design and synthesis. Whether you require sequence optimization, specific modifications, or larger production quantities, our team can support your project. Submit your project details and we will provide a personalized proposal.
References
07 September 2022
Cytokinesis Blocked Micronuclei Aberration Analysis
Abstract
Cytokinesis blocked micronuclei (CBMN) assay is a rapid and sensitive analysis of chromosome aberrations and miss assortments during cell division. Genotoxic agent exposure produces DNA damage and chromosome fragments. Fragmented chromosomes without centromere failed to attach kinetochore which segregates a pair of homologous chromosomes to each daughter cells at cytokinesis, hence leading to form micronuclei. Chromosome or fragments of chromosome can also form micronuclei when they are not accurately sorted to daughter cells. Using cytochalasin B, an actin inhibitor, blocks cytokinesis of which completion leads serration appearance formed with two daughter cells while nuclei segregation is undergoing. As a result, one cell having two daughter nuclei, i.e., binucleated cell, is produced. By analyzing these binucleated cells, chromosome aberrations can be estimated as well as popular chromosome aberration analysis. Frequency of micronuclei formation predicts the testing agents’ genotoxicity. By combining use with centromere-specific probes or DNA damage signal probes, the nature of genotoxicity of tested agents can be estimated.
34(6):p 829-836, June 2010. | DOI: 10.1097/PAS.0b013e3181dcede7
Shortened Telomeres in Serous Tubal Intraepithelial Carcinoma: An Early Event in Ovarian High-grade Serous Carcinogenesis
Abstract
Short telomeres are one of the main genetic manifestations in human cancer, as they have been shown to play an important role in inducing chromosomal instability and in contributing to tumor progression. The purpose of this study was to determine if changes in telomere length occur in serous tubal intraepithelial carcinoma (STIC), the putative precursor of “ovarian” high-grade serous carcinoma (HGSC). Twenty-two STICs from 15 patients with concurrent but discrete HGSCs were analyzed for telomere length on formalin-fixed, paraffin-embedded sections by conducting p53 immunofluorescence to assist in identifying STICs and telomere-specific FISH. Telomere length (short, long, or no change) in STICs was compared with HGSCs using normal fallopian tube epithelium and stromal cells as controls. We found that STICs had the shortest telomeres, as 18 (82%) of 22 STICs had short telomeres, whereas only 2 (9%) showed no change and 2 (9%) had long telomeres compared with the normal-looking tubal epithelium. In contrast, among 12 paired HGSCs and STICs, 6 HGSCs showed an increase in telomere length, one showed a decrease in length and 5 did not show any change when compared with their matched STICs, although, such as STICs, the majority of HGSCs had shorter telomeres than the associated normal tubal epithelial cells. These differences in telomere length between normal tubal epithelial cells and STICs, and between STICs and HGSCs were statisticaly significant (P<0.05). In conclusion, the finding of short telomeres, which have been shown to be one of the earliest molecular changes in carcinogenesis, in a vast majority of STICs provides further support to the proposal that STICs are precursors of HGSC and opens new areas of research in elucidating the early events of ovarian high-grade serous carcinogenesis.
Modern Pathology volume 25, pages1033–1039 (2012)
Loss of ATRX or DAXX expression and concomitant acquisition of the alternative lengthening of telomeres phenotype are late events in a small subset of MEN-1 syndrome pancreatic neuroendocrine tumors
Abstract
Approximately 45% of sporadic well-differentiated pancreatic neuroendocrine tumors harbor mutations in either ATRX (alpha thalassemia/mental retardation X-linked) or DAXX (death domain-associated protein). These novel tumor suppressor genes encode nuclear proteins that interact with one another and function in chromatin remodeling at telomeric and peri-centromeric regions. Mutations in these genes are associated with loss of their protein expression and correlate with the alternative lengthening of telomeres phenotype. Patients with multiple endocrine neoplasia-1 (MEN-1) syndrome, genetically defined by a germ line mutation in the MEN1 gene, are predisposed to developing pancreatic neuroendocrine tumors and thus represent a unique model for studying the timing of ATRX and DAXX inactivation in pancreatic neuroendocrine tumor development. We characterized ATRX and DAXX protein expression by immunohistochemistry and telomere status by telomere-specific fluorescence in situ hybridization in 109 well-differentiated pancreatic neuroendocrine lesions from 28 MEN-1 syndrome patients. The study consisted of 47 neuroendocrine microadenomas (<0.5 cm), 50 pancreatic neuroendocrine tumors (≥0.5 cm), and 12 pancreatic neuroendocrine tumor lymph node metastases. Expression of ATRX and DAXX was intact in all 47 microadenomas, and none showed the alternative lengthening of telomeres phenotype. ATRX and/or DAXX expression was lost in 3 of 50 (6%) pancreatic neuroendocrine tumors. In all three of these, tumor size was ≥3 cm, and loss of ATRX and/or DAXX expression correlated with the alternative lengthening of telomeres phenotype. Concurrent lymph node metastases were present for two of the three tumors, and each metastasis displayed the same changes as the primary tumor. These findings establish the existence of ATRX and DAXX defects and the alternative lengthening of telomeres phenotype in pancreatic neuroendocrine tumors in the context of MEN-1 syndrome. The observation that ATRX and DAXX defects and the alternative lengthening of telomeres phenotype occurred only in pancreatic neuroendocrine tumors measuring ≥3 cm and their lymph node metastases suggests that these changes are late events in pancreatic neuroendocrine tumor development.
